Total Dietary Fiber Assay Kit (100 Assays per Kit) (RINTDF)
Part #
The Rapid Integrated Total Dietary Fiber Assay Kit method is validated under collaborative study (AOAC Method 2017.16, ICC Standard No. 185) and suitable for the measurement and analysis of Total Dietary Fiber as per Codex Alimentarius definition.
This method is updated to be more consistent with in vivo conditions in the human small intestine, i.e. a 4 h incubation time. Under these conditions more accurate measurement of resistant starch is obtained, including phosphate cross-liked starch (RS4). Use of higher enzyme concentrations ensures that resistant maltodextrins produced from non-resistant starch under the incubation conditions of the Integrated Total Dietary Fiber procedure (AOAC Methods 2009.01 and 2011.25) are no longer produced.
In this improved, rapid method, the incubation time with PAA + AMG is reduced to 4 h and the levels of both PAA and AMG are increased to ensure that resistant starch levels obtained with a set of control samples are consistent with ileostomy data. Under these conditions, the DF values obtained for most samples are the same as those obtained with AOAC Methods 2009.01 and 2011.25.
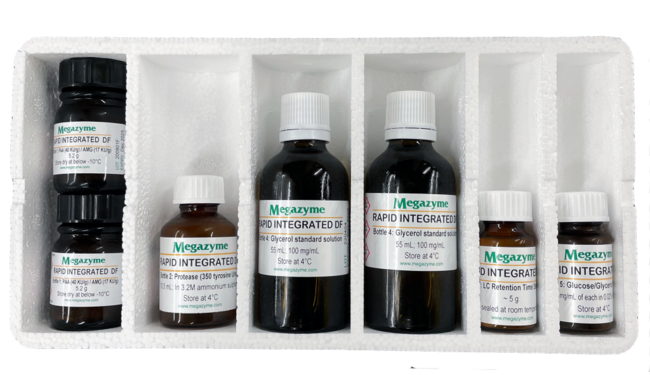
Able to support about 100 assays, the kit includes:
- Bottle 1 (Count 2), PAA (40 KU/g) / AMG (17 KU/g), 5.2 g
- Bottle 2 (Count 1), Protease (350 tyrosine U/ml, In 3.2 M ammonium sulphate), 10.5 ml
- Bottle 3 (Count 1), LC Retention Time Standard, ~ 5 g
- Bottle 4 (Count 2), Glycerol standard solution (100 mg/ml), 55 ml
- Bottle 5 (Count 1), Glucose / Glycerol standard (10 mg/ml of each in 0.02% sodium azide), 5 ml